Phase I Trial Publication
Safety and immunogenicity of the ChAdOx1 nCoV-19 vaccine against SARS-CoV-2:
a Phase I/2 randomized control trial
Summary
The Phase 1 data for our coronavirus vaccine shows that the vaccine raised no safety concerns, did not lead to any unexpected symptoms in those who received it, and was very similar to other vaccines of this type.
The immune responses that were made following vaccination are exactly the kind of responses that we think might be associated with protection, though we must continue with our rigorous clinical trial programme to confirm whether the responses we have seen are sufficient. Importantly, we saw an even greater immune response in participants who received two doses of the vaccine, indicating that this might be a good strategy for vaccination.
These encouraging results support large scale evaluation of this candidate vaccine in an ongoing Phase III programme, that is still needed to prove the vaccine will protect people from the virus.
Aims
The outcomes reported at this stage of the study include the safety of the vaccine and the immune responses of participants following vaccination.
The main objectives of the trial, including whether the vaccine can prevent COVID-19 disease will be reported at a later date.
The Trial
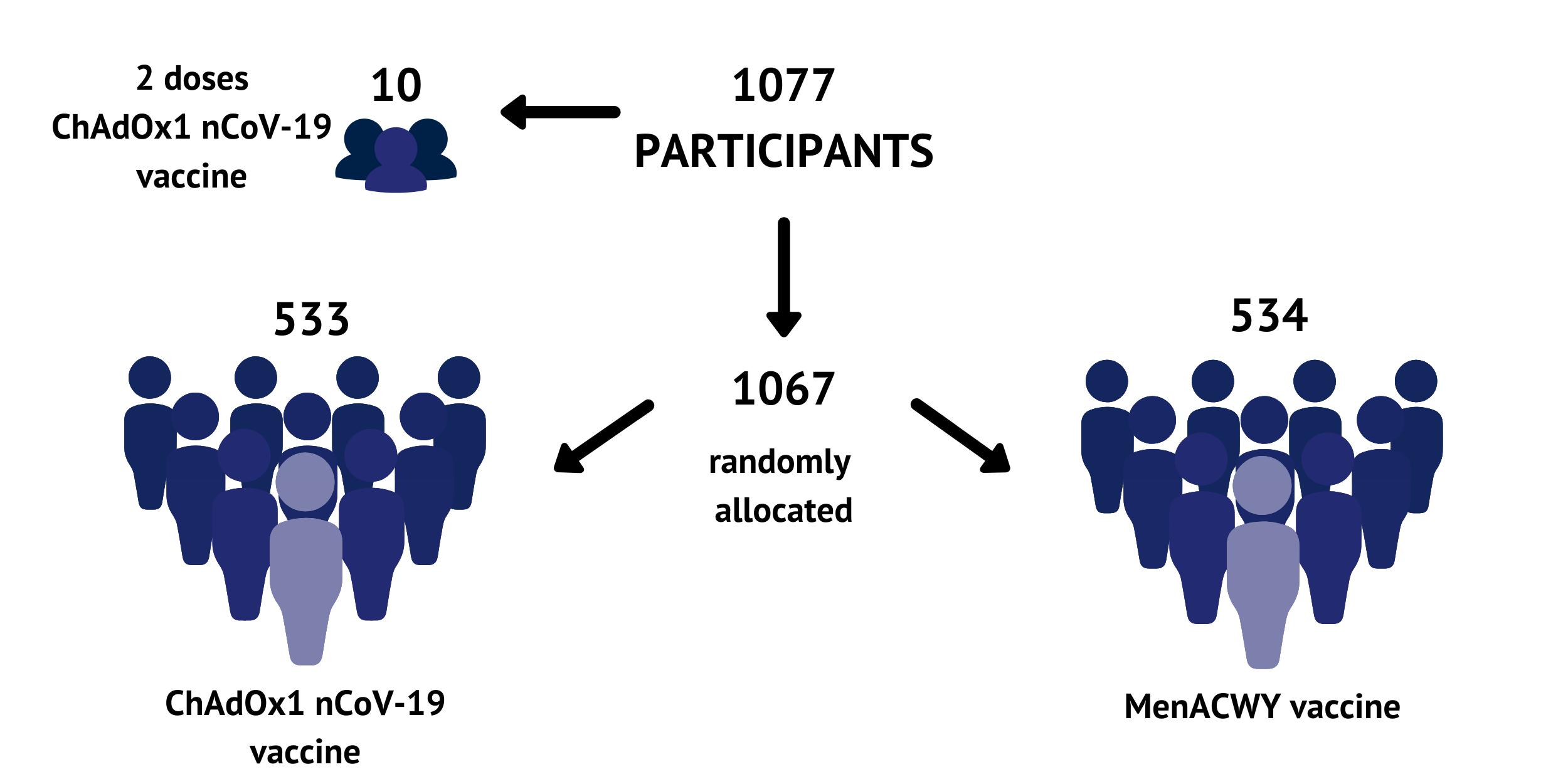
1077 participants have been recruited across multiple study sites in Oxford, Southampton, London and Bristol. Participants were randomly allocated to receive either the ChAdOx1 nCoV-19 vaccine or a licensed vaccine (MenACWY) that is used as a ‘control’ for comparison.
There was also a separate small group of 10 volunteers who received 2 doses of ChAdOx1 nCoV-19 four weeks apart. The dose used in this trial was chosen based on previous experiences with other ChAdOx1 based vaccines. All the participants are healthy adults between the ages of 18 and 55, 50% female and 50% male.
After receiving the vaccine, participants recorded any symptoms experienced for 7 days. They also recorded if they felt unwell for the following three weeks. Following the vaccination visit, participants attended a series of follow-up visits to check their observations and take blood samples. These blood samples were used to assess the immune response to the vaccine, by testing neutralising antibody and T cell responses.
Study participants will not know whether they received the ChAdOx1 nCoV-19 vaccine until the end of the trial.
The Results:
Safety
There were no safety concerns associated with the vaccine and no unexpected symptoms shown in those participants who received it. The safety profile was similar to that of other vaccines of this type, and included pain and tenderness at the injection site, and flu-like symptoms such as headache, fever and muscle aches. These symptoms were mainly reported in the first day following vaccination and were improved in individuals who received paracetamol for 24 hours from the time of vaccination. No severe adverse events associated with the ChAdOx1 nCoV-19 vaccine were reported.
Immunogenicity – Antibodies
Antibodies play an important role in the immune response to viruses. This study assessed both the quantity and quality of antibody found in participant blood samples, compared with samples from COVID-19 patients. An ELISA assay was used to measure the quantity of antibodies in the blood that recognise the spike protein. Following a single vaccination all participants produced spike specific antibody by 14 days, with the antibody response peaking at 28 days. This response was well maintained up to 56 days post vaccination showing potential for long term maintenance of the antibody response. In participants who received a second boost vaccination at 28 days, there was a further increase in the peak response. The levels of responses observed in vaccinated individuals are comparable to those seen in recovering COVID-19 patients.
As well as measuring the quantity of antibody, the study also examined the quality of antibody, its ability to neutralise the virus. Two types of neutralisation assay were used, one using live SARS-CoV-2 virus and another using a surrogate virus. As there is not currently a gold standard neutralisation assay, the team used multiple methods to assess the neutralising capacity of the antibody found in participant samples. Whilst these methods are not directly comparable to each other, or to those used in other vaccine development projects, they provide a broad assessment of this vaccine.
In combination, the neutralisation assay data suggests the following: By 28 days following a single vaccination, 91% of total volunteers had serum antibody that could neutralise the virus in one type of assay, and 37% had serum antibody that could neutralise the virus in a different, much more stringent assay. In the participants receiving a second dose of vaccine, 100% of volunteers were shown to have serum antibody capable of neutralising the virus using the more stringent assay. Whilst the data suggests a 2-dose regime may be superior to a single vaccine regime, it is still possible that the level of neutralising antibodies produced after a single dose may be sufficient for protection alongside a robust T cell response.
As we are still conducting long term follow up of participants in the study, data is not yet available on how long antibody lasts, however previous data using the same ChAdOx1 vaccine platform suggest this should be long lived (more than 1 year).
Immunogenicity – T Cells
T cells play an important role in the immune response to viral infections. Some T cells are responsible for killing viruses inside infected cells, whilst others are responsible for providing help to other components of the immune response. There is increasing evidence that T cells play an important role in preventing serious disease with natural infection of the COVID-19 virus.
T cell numbers were measured using the ELISpot assay. Following vaccination, the number of T cells in the blood of participants increased and peaked at 14 days post vaccination. This response dropped slightly by 28 days, but is then well maintained up to one month after vaccination. In contrast, T cell responses of participants in the control group were unchanged over the duration of the study. In the boost group, where participants received a second vaccination 4 weeks after the first, there was no further increase in T cells. These responses are as expected for this type of vaccine.
What’s next?
The participants in this study continue to be monitored to assess how well the immune responses are maintained over a longer time period.
If participants develop COVID-19 symptoms during the study, they can contact a member of the clinical team, to check whether they have become infected with the virus.
To assess whether the vaccine works to protect from COVID-19, the statisticians in our team will compare the number of infections in the control group with the number of infections in the vaccinated group. How quickly we reach the numbers required will depend on the levels of virus transmission in the community. With the current low transmission levels in the UK, this could take many months.
We have so far recruited over 8000 people to our Phase II/III study which aims to assess the efficacy of the vaccine, and to compare the different dose schedules. To increase the chance of achieving an efficacy result sooner, we are prioritising those who have a higher chance of being exposed to the SARS-CoV-2 virus, such as frontline healthcare workers, frontline support staff and public-facing key workers. Conducting studies in multiple locations where transmission levels vary also increases the chances of reaching an efficacy endpoint sooner.
The full publication is available here:




